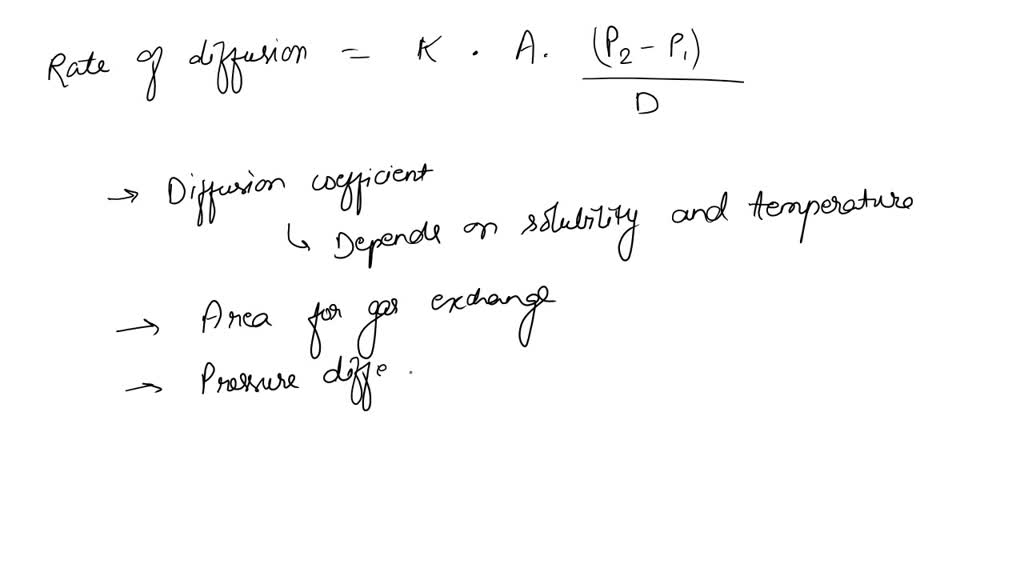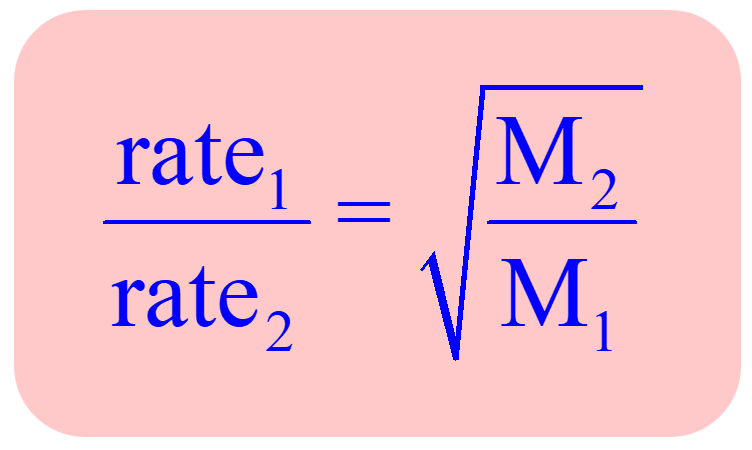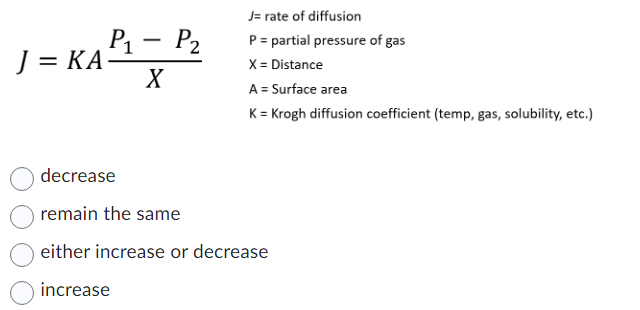The relative rate of effusion of ch4 to so2 through the container containing ch4 and so2 in 3:2mass ratio

The rate of diffusion of methane a given temperature is twice of a gas X . The molar mass of the gas X is?

Graham's Law of Diffusion Graham's Law KE = ½mv 2 Speed of diffusion/ effusion –Kinetic energy is determined by the temperature of the gas. –At the same. - ppt download
Can the rate of effusion or diffusion be negative, in accordance with Graham's law? If so, how? - Quora